|
Foodlaw-Reading
Dr David Jukes, The University of
Reading, UK
Providng access to food law since May 1996 |

|
.....  ..... ..... ..... .....  ..... .....  ..... .....  ..... ..... 
|
Last update
18 January, 202324 February, 2023ial, Helvetica, sans-serif">Food for Specific Groups
Providing access to EU and UK legislation
Summary
The first controls on foods for particular nutritional uses were introduced in 1977 by Directive 77/94. This created the concept of 'foods for particular nutritional uses' (commonly referred to as PARNUTS) and defined the concept in two ways. It stated that: "Foodstuffs for particular nutritional uses are foodstuffs which, owing to their special composition or manufacturing process , are clearly distinguishable from foodstuffs for normal consumption, which are suitable for their claimed nutritional purposes and which are marketed in such a way as to indicate such suitability." It then added that: "A particular nutritional use must fulfil the particular nutritional requirements: (i) of certain categories of persons whose digestive processes or metabolism are disturbed, or (ii) of certain categories of persons who are in a special physiological condition and who are therefore able to obtain special benefit from a controlled consumption of certain substances in foodstuffs, or (iii) of infants or young children in good health." It also reserved the use of the words 'dietetic' and 'dietary' to foods meeting these definitions.
As part of the process to create the 'Internal Market', the legislation was modified in 1989 with the adoption of Directive 89/398. This created a 'framework' control (containing the same elements as Directive 77/94) and provisions for the adoption of various 'specific Directives' for products listed in an Annex. The original list contained the following:
1. Infant formulae; 2. Follow-up milk and other follow-up foods; 3. Baby foods; 4. Low-energy and energy-reduced foods intended for weight control; 5. Dietary foods for special medical purposes; 6. Low-sodium foods, including low-sodium or sodium-free dietary salts; 7. Gluten-free foods; 8. Foods intended to meet the expenditure of intense muscular effort, especially for sportsmen; 9. Foods for persons suffering from carbohydrate-metabolism disorders (diabetes)
The need for all these specific controls was subject to some discussion and in 1999 (by Directive 99/41) the list was reduced to the following (with the possibility of also including diabetic foods still left as an option):
1. Infant formulae and follow-on formulae; 2. Processed cereal-based foods and baby foods for infants and young children; 3. Food intended for use in energy-restricted diets for weight reduction; 4. Dietary foods for special medical purposes; 5. Foods intended to meet the expenditure of intense muscular effort, especially for sportsmen
A new text, with limited changes, was subsequently published in 2009 (Directive 2009/39). However in 2011 the Commission proposed a simplified structure limiting controls to 4 main categories. This was subsequently adopted in 2013 as Regulation No 609/2013. This removed the concept of 'dietetic' or 'dietary' foods and established the framework for specific controls on the following:
(a) infant formula and follow-on formula; (b) processed cereal-based food and baby food; (c) food for special medical purposes; (d) total diet replacement for weight control.
It did also include the option of specific controls for 'Milk-based drinks and similar products intended for young children' and 'Food intended for sportspeople'. Controls on the four categories exist (see below). Following Commission reports on the 2 optional categories, it is unlikely that that these will be progressed. The title of the new Regulation is long and refers to the various categories to which it applies. The simplified term, 'Food for Specific Groups' has therefore been informally adopted to cover these terms and to replace the original PARNUTS acronym. The Regulation became effective from 20 July 2016.
The following illustrates some aspects of this chronology:
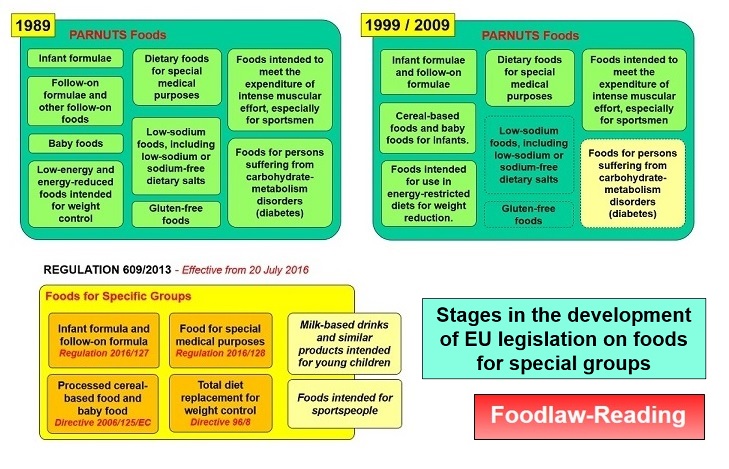
For a larger version of this figure, see: Figure: Foods for Special Groups - History (pdf format)
For the Commission's page on this topic, see: Foods for Specific Groups.
EU Legislation
Main Controls
- Regulation (EU) No 609/2013 of the European Parliament and of the Council of 12 June 2013 on food intended for infants and young children, food for special medical purposes, and total diet replacement for weight control and repealing Council Directive 92/52/EEC, Commission Directives 96/8/EC, 1999/21/EC, 2006/125/EC and 2006/141/EC, Directive 2009/39/EC of the European Parliament and of the Council and Commission Regulations (EC) No 41/2009 and (EC) No 953/2009 (OJ L181, 29.6.2013, page 35)
Amendments:
- Commission Delegated Regulation (EU) 2017/1091 of 10 April 2017 amending the Annex to Regulation (EU) No 609/2013 of the European Parliament and of the Council as regards the list of substances that may be added to processed cereal-based food and baby food and to food for special medical purposes (OJ L158, 21.6.2017, page 5)
- Commission Delegated Regulation (EU) 2021/571 of 20 January 2021 amending the Annex to Regulation (EU) No 609/2013 of the European Parliament and of the Council as regards the list of substances that may be added to infant and follow-on formula, baby food and processed cereal-based food (OJ L120, 8.4.2021, page 1)
- Commission Delegated Regulation (EU) 2023/439 of 16 December 2022 amending the Annex to Regulation (EU) No 609/2013 of the European Parliament and of the Council to allow the use of nicotinamide riboside chloride as a source of niacin in food for special medical purposes and total diet replacement for weight control (OJ L 64, 1.3.2023, page 1)
Specific Controls
Infant formula and follow-on formula
- Commission Delegated Regulation (EU) 2016/127 of 25 September 2015 supplementing Regulation (EU) No 609/2013 of the European Parliament and of the Council as regards the specific compositional and information requirements for infant formula and follow-on formula and as regards requirements on information relating to infant and young child feeding (OJ L25, 2.2.2016, page 1)
as amended by:
- Commission Delegated Regulation (EU) 2019/828 of 14 March 2019 amending Delegated Regulation (EU) 2016/127 with regard to vitamin D requirements for infant formula and erucic acid requirements for infant formula and follow-on formula (OJ L137, 23.5.2019, page 12)
- Commission Delegated Regulation (EU) 2021/572 of 20 January 2021 amending Delegated Regulation (EU) 2016/127 as regards the date of application of certain of its provisions (OJ L120, 8.4.2021, page 4)
- Commission Delegated Regulation (EU) 2021/1041 of 16 April 2021 amending Delegated Regulation (EU) 2016/127 as regards the requirements on pesticides in infant formula and follow-on formula (OJ L225, 25.6.2021, page 4)
- Commission Delegated Regulation (EU) 2022/519 of 14 January 2022 amending Delegated Regulation (EU) 2016/127 as regards the protein requirements for infant and follow-on formula manufactured from protein hydrolysates (OJ L 104, 1.4.2022, page 58)
- Commission Delegated Regulation (EU) 2023/589 of 10 January 2023 amending Delegated Regulation (EU) 2016/127 as regards the protein requirements for infant and follow-on formula manufactured from protein hydrolysates (OJ L 79, 17.3.2023, page 40)
Processed cereal-based food and baby food
- Commission Directive 2006/125/EC of 5 December 2006 on processed cereal-based foods and baby foods for infants and young children (OJ L339, 6.12.2006, page 16) [Note: this Directive is expected to be replaced with an updated Regulation in the near future]
Food for special medical purposes
- Commission Delegated Regulation (EU) 2016/128 of 25 September 2015 supplementing Regulation (EU) No 609/2013 of the European Parliament and of the Council as regards the specific compositional and information requirements for food for special medical purposes (OJ L25, 2.2.2016, page 30) as amended by:
- Commission Delegated Regulation (EU) 2021/1040 of 16 April 2021 amending Delegated Regulation (EU) 2016/128 as regards the requirements on pesticides in food for special medical purposes developed to satisfy the nutritional requirements of infants and young children (OJ L225, 25.6.2021, page 1)
For previous controls, see below
Total diet replacement for weight control
- Commission Delegated Regulation (EU) 2017/1522 of 2 June 2017 supplementing Regulation (EU) No 609/2013 of the European Parliament and of the Council as regards the specific compositional and information requirements for total diet replacement for weight control (OJ L230, 6.9.2017, page 1)
- Corrigendum to Commission Delegated Regulation (EU) 2017/1522 of 2 June 2017 supplementing Regulation (EU) No 609/2013 of the European Parliament and of the Council as regards the specific compositional and information requirements for total diet replacement for weight control (OJ L 230, 6.9.2017) (OJ L234, 12.9.2017, page 7) [Note: This Corrigendum states: 'The publication of Commission Delegated Regulation (EU) 2017/1522 is to be considered null and void.']
- Commission Delegated Regulation (EU) 2017/1798 of 2 June 2017 supplementing Regulation (EU) No 609/2013 of the European Parliament and of the Council as regards the specific compositional and information requirements for total diet replacement for weight control (OJ L259, 7.10.2017, page 2) as amended by:
- Commission Delegated Regulation (EU) 2022/2182 of 30 August 2022 amending Delegated Regulation (EU) 2017/1798 as regards the lipid and magnesium requirements for total diet replacement for weight control (OJ L 288, 9.11.2022, page 18)
Selected Links to Earlier EU Legal Documents
Food for special medical purposes
- Commission Directive 1999/21/EC of 25 March 1999 on dietary foods for special medical purposes
(OJ L91, 7.4.1999, page 29)
- Corrigendum to the Commission Directive 1999/21/EC on dietary foods for special medical purposes (OJ L 91 of 7.4.1999)
(OJ L2, 5.1.2000, page 79)
Amendments:
- Commission Directive 2006/82/EC of 23 October 2006 adapting Directive 91/321/EEC on infant formulae and follow-on formulae and Directive 1999/21/EC on dietary foods for special medical purposes, by reason of the accession of Bulgaria and Romania (OJ L362, 20.12.2006, page 94)
- Commission Directive 2006/141/EC of 22 December 2006 on infant formulae and follow-on formulae and amending Directive 1999/21/EC (OJ L401, 30.12.2006, page 1)
- Commission Directive 2013/26/EU of 8 February 2013 adapting certain directives in the field of food safety, veterinary and phytosanitary policy, by reason of the accession of Croatia (OJ L158, 10.6.2013, page 376)
A consolidated text is available. See: Directive 1999/21 - Consolidated Text (July 2013)
Energy-restricted diets for weight reduction
- Commission Directive 96/8/EC of 26 February 1996
on foods intended for use in energy-restricted diets for weight reduction (OJ L55, 6.3.1996, page 22)
Amendment:
- Commission Directive 2007/29/EC of 30 May 2007 amending Directive 96/8/EC as regards labelling, advertising or presenting foods intended for use in energy-restricted diets for weight reduction (OJ L139, 315.2007, page 2)
UK Legislation and Guidance
Brexit: Prior to the IP Completion Day (31 December 2020), the legal requirements given in the EU Regulations listed above still applied to the UK. Since IP Completion Day, the EU Regulations above have been incorporated into UK legislation but with amendments to correct deficiencies. Information on this is given below. For more details of the process of incorporating EU legislation into UK law, see the separate page: UK Food Law: EU Legislation as Amended for the UK. Provisions for the enforcement of the controls (originally the EU Regulations but now as amended) have been provided in the UK Regulations listed below. For Northern Ireland, EU rules still apply.
1st January 2021: For guidance, see:
EU Legislation amended for application in the UK: with links to legislation.gov.uk
- Regulation (EU) No 609/2013 of the European Parliament and of the Council of 12 June 2013 on food intended for infants and young children, food for special medical purposes, and total diet replacement for weight control ... as amended by:
- Nutrition (Amendment etc.) (EU Exit) Regulations 2019 (SI 2019, No. 651) as amended by:
- Nutrition (Amendment etc.) (EU Exit) Regulations 2020 (SI 2020, No. 1476) as amended by:
- Nutrition (Amendment) and Food for Specific Groups (Food for Special Medical Purposes for Infants, Infant Formula and Follow-on Formula) (Information and Compositional Requirements) (Amendment) Regulations 2021 (SI 2021, No. 168)
- Food Supplements and Food for Specific Groups (Miscellaneous Amendments) Regulations 2023 (SI 2023, No. 28) as amended by:
- Food Supplements and Food for Specific Groups (Miscellaneous Amendments) (No. 2) Regulations 2023 (SI 2023, No. 131)
- Commission Delegated Regulation (EU) 2016/127 of 25 September 2015 supplementing Regulation (EU) No 609/2013 of the European Parliament and of the Council as regards the specific compositional and information requirements for infant formula and follow-on formula as amended by:
- Nutrition (Amendment etc.) (EU Exit) Regulations 2020 (SI 2020, No. 1476) as amended by:
- Nutrition (Amendment) and Food for Specific Groups (Food for Special Medical Purposes for Infants, Infant Formula and Follow-on Formula) (Information and Compositional Requirements) (Amendment) Regulations 2021 (SI 2021, No. 168)
- Food Supplements and Food for Specific Groups (Miscellaneous Amendments) Regulations 2023 (SI 2023, No. 28) as amended by:
- Food Supplements and Food for Specific Groups (Miscellaneous Amendments) (No. 2) Regulations 2023 (SI 2023, No. 131)
- Commission Delegated Regulation (EU) 2016/128 of 25 September 2015 supplementing Regulation (EU) No 609/2013 of the European Parliament and of the Council as regards the specific compositional and information requirements for food for special medical purposes as amended by:
- Nutrition (Amendment etc.) (EU Exit) Regulations 2019 (SI 2019, No. 651) as amended by:
- Nutrition (Amendment etc.) (EU Exit) Regulations 2020 (SI 2020, No. 1476) as amended by:
- Nutrition (Amendment) and Food for Specific Groups (Food for Special Medical Purposes for Infants, Infant Formula and Follow-on Formula) (Information and Compositional Requirements) (Amendment) Regulations 2021 (SI 2021, No. 168)
- Food Supplements and Food for Specific Groups (Miscellaneous Amendments) Regulations 2023 (SI 2023, No. 28) as amended by:
- Food Supplements and Food for Specific Groups (Miscellaneous Amendments) (No. 2) Regulations 2023 (SI 2023, No. 131)
- Commission Delegated Regulation (EU) 2017/1798 of 2 June 2017 supplementing Regulation (EU) No 609/2013 of the European Parliament and of the Council as regards the specific compositional and information requirements for total diet replacement for weight control [Note: will only apply from 27 October 2022. Currently subject to UK Regulations based on the EU Directive]
UK Legislation based on EU Directives
Processed cereal-based food and baby food (implenting Commission Directive 2006/125/EC)
- Processed Cereal-based Foods and Baby Foods for Infants and Young Children (England) Regulations 2003 (SI 2003, No. 3207) as amended by
- Food Supplements and Food for Specific Groups (Miscellaneous Amendments) Regulations 2023 (SI 2023, No. 28) as amended by:
- Food Supplements and Food for Specific Groups (Miscellaneous Amendments) (No. 2) Regulations 2023 (SI 2023, No. 131)
- Processed Cereal-based Foods and Baby Foods for Infants and Young Children (Wales) Regulations 2004 (SI 2004, No. 314 (W.32) as amended by
- Processed Cereal-based Foods and Baby Foods for Infants and Young Children (Wales) (Amendment) Regulations 2022 (SI 2022 No. 1327 (W.268))
- Processed Cereal-based Foods and Baby Foods for Infants and Young Children (Scotland) Regulations 2004 (SSI 2004, No. 8) as amended by:
- Processed Cereal-based Foods and Baby Foods for Infants and Young Children (Scotland) Amendment Regulations 2022 (SSI 2022, No. 342)
- Processed Cereal-based Foods and Baby Foods for Infants and Young Children Regulations (Northern Ireland) 2003 (SRNI 2003, No. 530)
[Note that the 2006 Directive was a codification of the earlier 1996 Directive as amended in 1998, 1999 and 2003]
Energy-restricted diets for weight reduction (implenting Commission Directive 96/8/EC)
- Foods
Intended for Use in Energy Restricted Diets for Weight Reduction Regulations
1997 (SI 1997, No 2182) [for E, W and S] as amended by:
- Official Feed and Food Controls (Scotland) Regulations 2005 (SSI 2005, No. 616) [for S]
- Official Feed and Food Controls (England) Regulations 2005 (SI 2005, No. 2626) [for E]
- Official Feed and Food Controls (Wales) Regulations 2005 (SI 2005, No. 3254 (W.247)) [for W]
- Food for Particular Nutritional Uses (Scotland) (Miscellaneous Amendments) Regulations 2007 (SSI 2007, No. 424) [for S]
- Food for Particular Nutritional Uses (Miscellaneous Amendments) (England) Regulations 2007 (SI 2007, No. 2591 [for E]
- Food for Particular Nutritional Uses (Miscellaneous Amendments) (Wales) Regulations 2007 (SI 2007, No. 2753 (W.232)) [for W]
- Food Information (Scotland) Regulations 2014 (SSI 2014, No. 312) [for S]
- Food Information Regulations 2014 (SI 2014, No. 1855) [for E]
- Food Information (Wales) Regulations 2014 (SI 2014, No. 2303 (W.227)) [for W]
- Foods for Specific Groups (Scotland) Regulations 2016 (SSI 2016, No. 190) [for S]
- Food for Specific Groups (Information and Compositional Requirements) (Wales) Regulations 2016 (SI 2016, No. 639 (W.175)) [for W]
- Food for Specific Groups (Information and Compositional Requirements) (England) Regulations 2016 (SI 2016, No. 688) [for E]
- Foods for Specific Groups (Medical Foods) (Miscellaneous Amendments) (Scotland) Regulations 2018 (SSI 2018, No. 392) [for S]
- Food and Feed (Miscellaneous Amendments and Revocations) (Wales) Regulations 2018 (SI 2018, No. 806 (W.162)) [for W]
- Food for Specific Groups (Food for Special Medical Purposes for Infants, Infant Formula and Follow-on Formula) (Information and Compositional Requirements) (Amendment etc.) (England) Regulations 2020 (SI 2020, No. 43) [for E] as amended by:
- Nutrition (Amendment) and Food for Specific Groups (Food for Special Medical Purposes for Infants, Infant Formula and Follow-on Formula) (Information and Compositional Requirements) (Amendment) Regulations 2021 (SI 2021, No. 168)
- Foods Intended for Use in Energy Restricted Diets for Weight Reduction Regulations (Northern Ireland) 1997 (SRNI 1997, No 450) [NI] as amended by:
- Official Feed and Food Controls Regulations (Northern Ireland) 2005 (SRNI 2005, No. 574)
- Food for Particular Nutritional Uses (Miscellaneous Amendments) Regulations (Northern Ireland) 2007 (SRNI 2007, No. 408)
- Food Information Regulations (Northern Ireland) 2014 (SRNI 2014, No. 223)
- Food Safety (Information and Compositional Requirements) Regulations (Northern Ireland) 2016 (SRNI 2016, No. 251)
Enforcement
Requirements for implementation and enforcement of the requirements of EU Regulations are provided separately for the four parts of the United Kingdom.
- England
- Food for Specific Groups (Information and Compositional Requirements) (England) Regulations 2016 (SI 2016, No. 688) [Revised as amended]
- Food for Specific Groups (Food for Special Medical Purposes for Infants, Infant Formula and Follow-on Formula) (Information and Compositional Requirements) (Amendment etc.) (England) Regulations 2020 (SI 2020, No 43) as amended by:
- Nutrition (Amendment) and Food for Specific Groups (Food for Special Medical Purposes for Infants, Infant Formula and Follow-on Formula) (Information and Compositional Requirements) (Amendment) Regulations 2021 (SI 2021, No. 168)
- Wales
- Food for Specific Groups (Information and Compositional Requirements) (Wales) Regulations 2016 (SI 2016, No 639 (W.175)) as amended by:
- Food for Specific Groups (Information and Compositional Requirements) (Amendment) (Wales) Regulations 2019 (SI 2019, No. 70 (W.22))
- Nutrition (Miscellaneous Amendments) (Wales) (EU Exit) Regulations 2019 (SI 2019, No. 179 (W.45))
as amended by:
- Infant Formula and Follow-on Formula (Wales) Regulations 2020 (SI 2020, No. 89 (W.13))
- Food for Specific Groups (Information and Compositional Requirements) (Wales) (Amendment) Regulations 2020 (SI 2020, No. 92 (W.14))
- Infant Formula and Follow-on Formula (Wales) Regulations 2020 (SI 2020, No. 89 (W.13))
as amended by:
- Scotland
- Foods for Specific Groups (Scotland) Regulations 2016
(SSI 2016, No. 190)
as amended by:
- Foods for Specific Groups (Medical Foods) (Miscellaneous Amendments) (Scotland) Regulations 2018 (SSI 2018, No. 392)
- Foods for Specific Groups (Medical Foods for Infants) and Addition of Vitamins, Minerals and Other Substances (Scotland) Amendment Regulations 2020 (SSI 2020, No. 7)
- Food and Feed (EU Exit) (Scotland) (Amendment) Regulations 2020 (SSI 2020, No. 372)
- Foods for Specific Groups (Infant Formula and Follow-on Formula) (Scotland) Regulations 2020 (SSI 2020, No. 6) [Infant formulae] as amended by:
- Foods for Specific Groups (Infant Formula and Follow-on Formula) (Scotland) Amendment Regulations 2021 (SSI 2021, No. 123)
- Northern Ireland
- Food Safety (Information and Compositional Requirements) Regulations (Northern Ireland) 2016 (SRNI 2016, No. 251)
as amended by:
- Food (Miscellaneous Amendments and Revocations) Regulations (Northern Ireland) 2019 (SRNI 2019, No. 5)
- Food Safety (Information and Compositional Requirements) (Amendment) Regulations (Northern Ireland) 2019 (SRNI 2019, No. 9)
- Food Safety (Information and Compositional Requirements) (Amendment) Regulations (Northern Ireland) 2020 (SRNI 2020, No 16)
- Food Safety (Information and Compositional Requirements) (Amendment) Regulations (Northern Ireland) 2020 (SRNI 2020, No 16) [Medical foods and Infant formulae]
Guidance
|
DHSC Guidance: Commission Delegated Regulation (EU) 2016/127 (supplementing Regulation (EU) No 609/2013) (Web page - Updated March 2022) |
 |
Department of Health: Guidance notes on the notification of marketing of foods for particular nutritional uses, medical foods and infant formula
Originally published in 2013. See also: Medical foods notification form (updated February 2019) |
|
Nutrition Legislation Information Sheet.
This document, published by the Department of Health and Social Care, is intended to help food businesses comply with nutrition legislation - some sections relate to food for specific groups. (Originally published in 2014, this is a pdf version of the updated DHSC web page from November 2022).
[Provided under the Open Government Licence. The original publication accessed from:  ] ] |
This page was first provided on 12 August 2016
To go to main Foodlaw-Reading
Index page, click here.
![]()

